All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MDS Alliance.
The MDS Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MDS Hub cannot guarantee the accuracy of translated content. The MDS Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View MDS content recommended for you
Referral patterns and potential barriers to transplantation in patients with MDS
Do you know... Which of the following are potential reasons a patient with high-risk myelodysplastic syndrome or acute myeloid leukemia would not be eligible for hematopoietic stem cell transplantation?
Hematopoietic stem cell transplantation (HSCT) can be a successful treatment for patients with high-risk myelodysplastic syndrome (HR-MDS) or acute myeloid leukemia (AML).1 However, factors such as age, performance status, comorbidities, and patient frailty may influence transplant referral patterns and outcomes in this population. Currently, the proportion of eligible patients being referred for transplantation is not known in clinical settings.1
The MDS Hub has previously covered the American Society for Transplantation and Cellular Therapy recommendations for transplantation in patients with MDS. Here we present a summary of the findings from a prospective cohort study on the patterns of consideration for HSCT in patients with HR-MDS and AML.1 In addition, we also summarize a retrospective study of survival benefits with allogeneic-HSCT (allo-HSCT) versus decitabine, a hypomethylating agent (HMA) agent presented by Runzhi at the European Hematology Association (EHA) 2023.2
Study design1
This analysis comprised data from the Connect Myeloid Disease Registry (NCT01688011), a multicenter, prospective, observational cohort study in patients with newly diagnosed MDS or AML.1 Cohorts included patients enrolled at community (CO), government (GOV), or academic (AC) sites. Eligible patients were:1
- Diagnosed with MDS or AML according to the 2008 World Health Organization criteria;
- ≥18 years of age if diagnosed with MDS or ≥55 years if diagnosed with AML; and
- Enrolled ≥60 days after a diagnosis of MDS or AML.
Patients were assessed for HSCT eligibility at baseline and at each follow-up visit. HSCT eligibility criteria included considered potentially eligible, not considered potentially eligible, or not assessed.
Results
Baseline characteristics
Overall, 778 patients were included, of which 66.3% were from the CO/GOV cohort and 33.7% from the AC cohort.1 Patients in the AC cohort were younger (p < 0.0001), more likely to reside in the Northeastern United States (p < 0.0001), and more likely to have a private insurance (p = 0.017) compared with the CO/GOV cohort (Table 1).
Table 1. Baseline characteristics*
|
AC, academic; AML, acute myeloid leukemia; AR, adverse risk; CO/GOV, community/government; HR, higher-risk; IR, immediate risk; MDS, myelodysplastic syndromes. |
||
|
Characteristics, % (unless otherwise specified) |
CO/GOV cohort (n = 516) |
AC cohort (n = 262) |
|---|---|---|
|
Median age, (range) years |
73 (19–97) |
69 (34–90) |
|
Sex, male |
64.1 |
62.6 |
|
Not specified |
8.7 |
5.7 |
|
Primary insurance† |
||
|
Medicare |
66.9 |
57.0 |
|
Private |
24.7 |
32.8 |
|
Medicaid |
1.6 |
3.5 |
|
Other |
6.9 |
6.6 |
|
Overall comorbidity grade |
||
|
0 or 1 |
50.8 |
59.0 |
|
2 or 3 |
49.2 |
41.0 |
|
Diagnosis† |
|
|
|
HR-MDS |
50.4 |
30.9 |
|
IR-AML |
30.2 |
36.6 |
|
AR-AML |
19.4 |
32.4 |
Transplantation rates and barriers to transplantation1
- Patients from CO/GOV cohort were less likely to be considered potentially eligible for transplantation compared with those from AC cohort (27.9% vs 43.9%; p < 0.001).
- Multivariate analysis showed that when adjusted for age (<65 vs ≥65 years) and comorbidity grade (<2 vs ≥2), patients in CO/GOV cohort were less likely to be considered eligible for transplantation compared with AC cohort (odds ratio, 1.6; p = 0.0155).
- Among patients eligible for transplantation, 45.1% from the CO/GOV cohort versus 35.7% from AC cohort underwent transplantation (p = 0.12).
- From the CO/GOV cohort 50.1% of patients were not considered eligible for transplantation versus 45.4% from the AC cohort. The main reasons for transplantation ineligibility are shown in Figure 1.
- The proportion of patients not assessed for transplantation was higher in the CO/GOV cohort compared with AC cohort (20.7% vs 10.7%, p = 0.0005).
Figure 1. Reasons for transplant ineligibility in CO/GOV versus AC cohort*
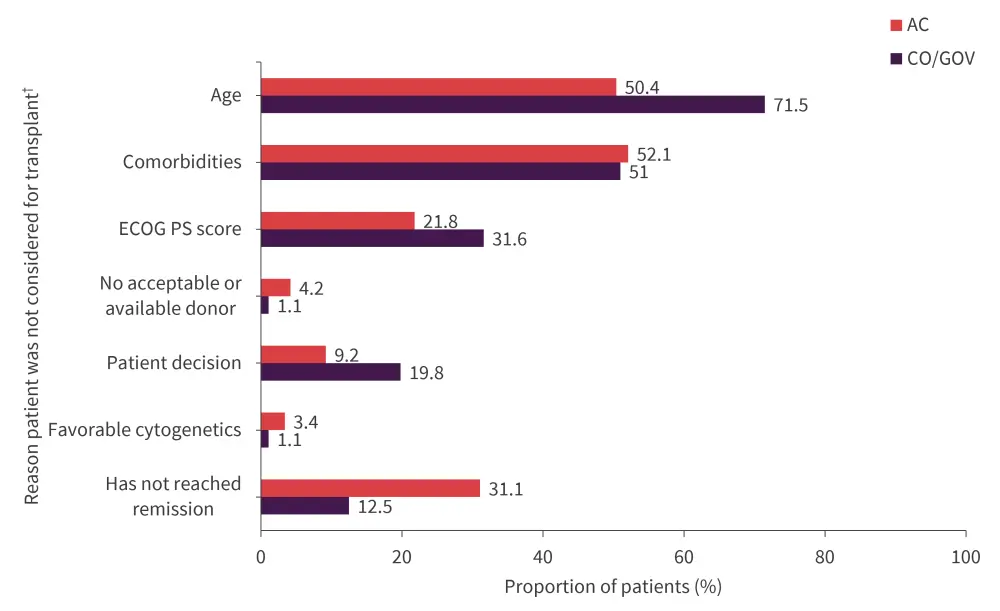
AC, academic; CO/GOV, community/government; ECOG PS, Eastern Cooperative Oncology Group performance status.
†≥1 is possible.
*Adapted from Tomlinson et al.1
Survival benefit of transplantation versus decitabine2
This was a retrospective study comparing responses to allo-HSCT with decitabine in patients with MDS with increased blasts type I (MDS-IBI). Outcomes included overall survival (OS), event-free survival (EFS), non-relapse mortality (NRM), and cumulative rate of relapse or progression (CIR) at 1 year.
Survival outcomes
- In total, 103 patients were assessed, with a median age of 49 years. Allogeneic HSCT was performed in 61.2% of patients, while decitabine was administered continuously to the remaining 38.8% of patients.
- Patients who underwent allogeneic HSCT versus decitabine treatment had higher OS and EFS rates at 3 years and non-relapse mortality rate after 1 year (Figure 2).
- Cumulative incidence of relapse and progression after 1 year was lower in patients who underwent allo-HSCT versus decitabine treatment.
Figure 2. Survival rates in patients with MDS-IBI treated with allogeneic-HSCT vs decitabine*
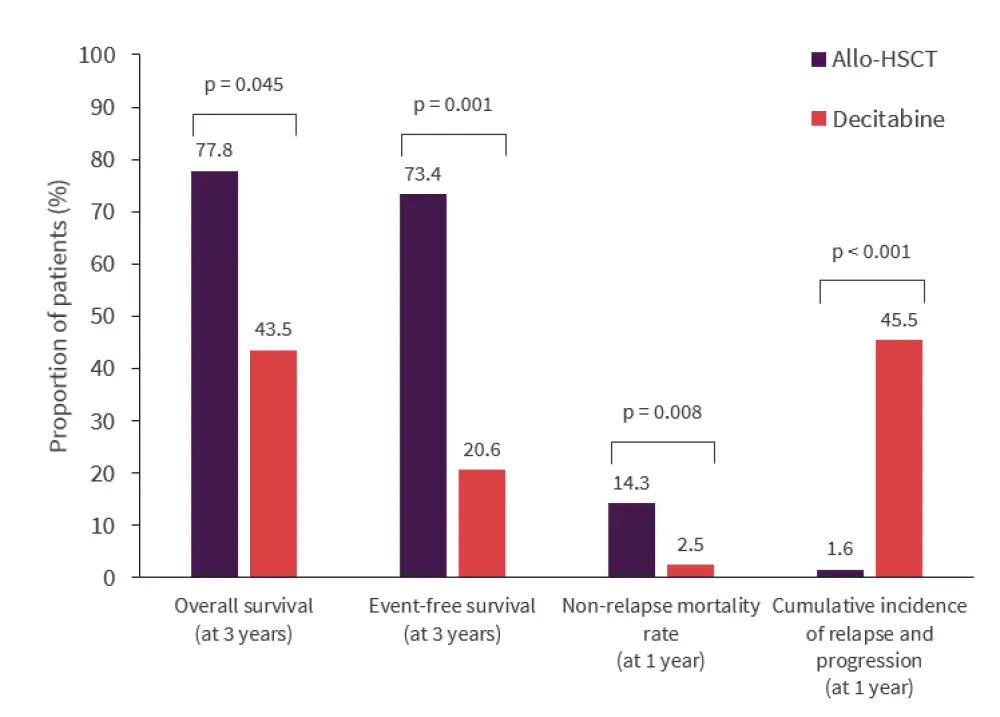
Allo-HSCT, allogeneic stem cell transplant; MDS-IBI, myelodysplastic syndrome with increased blasts type 1.
*Adapted from Runzhi.1
Conclusions
The study by Tomlinson1 showed that many patients who may be potential candidates for allo-HSCT are either not being assessed or considered for transplantation in clinical settings. This suggests the need for increasing resources for transplantation to ensure all patients are appropriately screened and referred for allo-HSCT if appropriate. The study by Runzhi2 demonstrates significantly prolonged survival in patients with MDS-IBI who underwent allogeneic-HSCT compared with those administered decitabine.
Together, both studies provide an insight into the success of allo-HSCT and how factors influencing eligibility should be considered in all patients, and patients should not be disregarded ahead of assessment for HSCT due to age and comorbidities.1,2
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

