All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MDS Alliance.
The MDS Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MDS Hub cannot guarantee the accuracy of translated content. The MDS Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View MDS content recommended for you
The IPSS-Molecular, a new risk stratification for patients with MDS
Your opinion matters
When it becomes available would you use the new IPSS-M calculator to risk stratify your patients?
Myelodysplastic syndromes (MDS) are a heterogeneous group of rare blood cancers and therefore have varying clinical outcomes. Some patients have mild disease and live relatively long, while others are affected more severely and may progress to acute myeloid leukemia. Therefore, it is crucial to identify the patients with a higher likelihood of progression and to treat them appropriately. The revised International Prognostic Scoring System (IPSS-R) is the main clinical algorithm used globally to inform both risk stratification and treatment decisions for patients with MDS. It considers the clinical features (hemoglobin levels, absolute neutrophil count, platelet count, percentage of bone marrow [BM] blasts), the five cytogenetic categories (very good, good, intermediate, poor, very poor), and age adjustment to stratify patients into one of five risk categories that can then inform subsequent treatment decisions. However, the IPSS-R does not currently include the use of molecular results. To address this, the International Working Group for the Prognosis of MDS (IWG-PM) have recently designed and validated a new risk scoring system—the IPSS-Molecular—that incorporates molecular diagnostic testing into the risk stratification.
The development and validation results for the IPPS-Molecular were presented by Elli Papaemmanuil1 during the 16th International Congress on Myelodysplastic Syndromes, and are summarized below.
Cohort characteristics
Overall, 3,324 patients with MDS with high quality peri-diagnostic samples and good clinical annotation were ascertained globally across 25 centers.
- All samples were assembled within Memorial Sloan Kettering Cancer Center to achieve uniform molecular profiling, across the genes most frequently mutated in MDS.
- 2,957 patients served as the training cohort for the development of the model.
- Analysis of the clinical variables showed that the cohort was highly representative of patients with MDS (Table 1).
Table 1. Clinical characteristics of the training cohort*
|
*Data adapted from Papaemmanuil E, 2021.1 |
|
|
Characteristic |
n = 2,957 |
|---|---|
|
Median follow-up, years |
3.8 |
|
Age at diagnosis, years (95th percentile range) |
72 (39–88) |
|
Median bone marrow blasts, % (95th percentile range) |
3 (0–16) |
|
Median hemoglobin, g/dL (range) |
10 (6–14) |
|
Platelet count ×109/L (range) |
130 (13–542) |
- 752 patients from the Japanese MDS Consortium were used as the validation cohort, which was enriched in higher-risk patients.
Results
Molecular landscape of the model cohort
- In terms of the proportion of patients in each category, the training cohort was representative of IPSS-R cytogenetic categories, IPSS-R risk scores, and World Health Organization 2016 categories.
- There were 3,073 cytogenetic alterations and 9,254 mutations mapped across 126 genes.
- Mapping of 9,189 mutations considered as oncogenic in 94 genes were seen in 0.2–31% of patients.
- The most affected gene was TET2, mutated in 31% of patients.
- These findings were consistent with previously published data.
- The number of driver events across the cohort varied, with ~6% of patients having no driver alterations by either gene mutations or cytogenetic findings.
- For patients with only one driver event, there was a notable enrichment in SF3B1, TET2, and deletion 5q mutations.
- The number of oncogenic driver events was also shown to be associated with increased severity in clinical presentation and risk.
- p < 0.0001 difference in the number of BM blasts
- p < 0.0001 difference in leukemia-free survival (LFS) probability
Encoding of clinical variables into IPSS-Molecular
The linear encoding of clinical features as continuous variables captured the risk representation for the percentage of BM blasts and hemoglobin levels. However, there was a cap in risk for platelet count, which occurred at 250 × 109/L.
- The absolute neutrophil count did not add any prognostic value; therefore, it was not included in the model.
Encoding of cytogenetic variables into IPSS-Molecular
- Comparison of individual cytogenetic events to IPSS-R cytogenetic risk categories did not improve model discrimination in terms of C-index for LFS; therefore, the IPSS-R cytogenetic categories were retained in the IPSS-Molecular.
- A significant improvement in model discrimination was seen with incorporation of gene mutations alone or gene mutations along with all other IPSS-R variables.
Encoding of genetic variables into the IPSS-Molecular
The correlation of specific genes with three endpoints—LFS, overall survival (OS) and AML-transformation (AML-t)—allowed for the evaluation of genes that should be included in the IPSS-Molecular.
- The top three mutations associated with adverse outcomes were
- bialleleic inactivation/multi-hit mutations in TP53;
- FLT3ITD+TKD (internal tandem duplications + tyrosine kinase domain); and
- MLLPTD (partial tandem duplication).
- SF3B1 remained the only gene associated with favorable outcomes, particularly as a negative predictor of AML transformation.
- Overall, 21 genes were found to correlate significantly with outcome and, therefore, should be included in the IPSS-Molecular: TP53mutli, MLLPTD, FLT3ITD/TKD, SF3B1, NPM1, RUNX1, IDH2, NRAS, ETV6, EZH2, SETBP1, CBL, SRSF2, U2AF1, DNMT3A, ASXL1, KRAS, NF1, STAG2, BCOR, and CEBPA.
Of note:
- Monoallelic TP53 mutations remained distinct from multi-hit mutations in TP53; therefore, the two types should be inputted separately into the IPSS-Molecular model.
- FLT3-ITD and -TKD mutations should be inputted together into the model.
- MLL-PTD analysis is not frequently ascertained on routine molecular panels for MDS as it only occurs in ~2.5% of patients.
Neither variant allele fraction estimates nor gene-to-gene interactions improved model discrimination in terms of C-index for LFS, so were not included in the model. However, as the number of driver events can have additive effects on outcome, the approach taken by the IWG-PM was to exclude the 21 genes already captured in the main model, but include those that were recurrent in the data set, were associated with adverse outcomes, and had additive risk effects.
- 17 genes were relevant for residual risk estimation: CSF3R, PTPN11, GATA2, ATRX, BCORL1, U2AF2, GNG1, ETNK1, CREBBP, PRPF8, WT1, ASXL2, LUC7L2, PPM1D, PHF6, SUZ12, and IDH1.
- Within the training cohort, most patients did not have any residual mutated genes (79%), and a small fraction had at most three residual mutated genes (Figure 1).
- Patients with two mutations in residual risk genes had a significantly shorter median LFS compared with those that did not (1.0 years vs 3.5 years).
Figure 1. Percentage of patients in the training cohort with residual mutated genes*
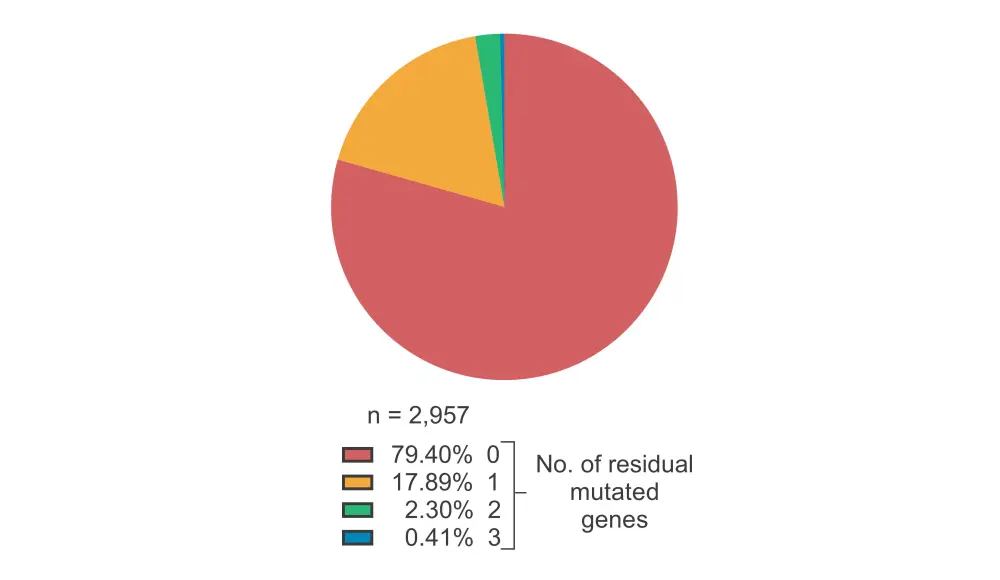
*Data adapted from Papaemmanuil E, 2021.1
Development of the patient-specific risk score
All four factors mentioned above (clinical features, cytogenetic features, gene mutations with main effects, and gene mutations with residual effects) were combined to develop a reproducible formula to derive patient-specific risk scores, necessary for the adoption of IPSS-Molecular into practical and interpretable clinical frameworks.
- The formula estimates a person’s risk score on a continuous scale based on a log 2 hazard ratio; therefore, an increase of one point in the IPSS-Molecular score indicates a doubling of a patient’s risk.
- Patients were separated into six risk strata: very high, high, moderate–high, moderate–low, low, and very low (Figure 2).
Figure 2. Percentage of patients across each strata in the IPSS-Molecular*
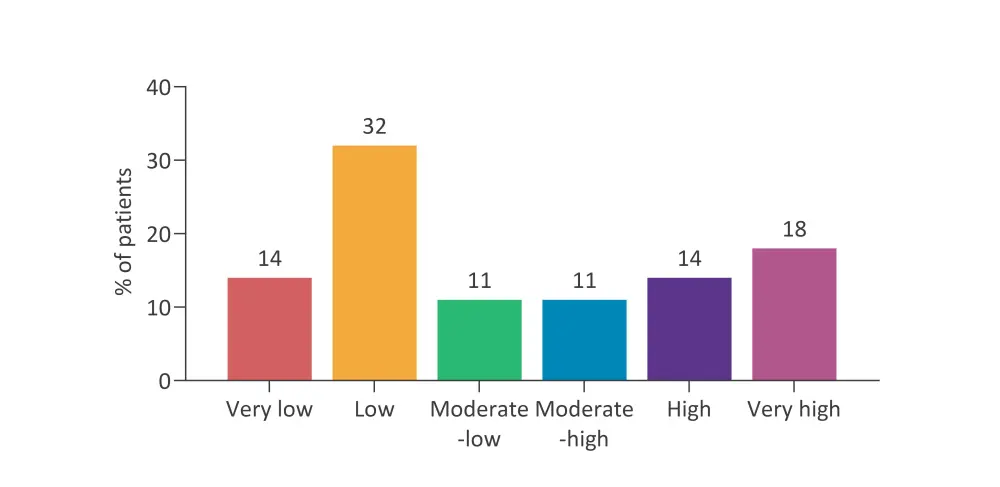
IPSS; International Prognostic Scoring System.
*Data adapted from Papaemmanuil E, 2021.1
- Each of the risk strata showed very good separation terms of OS (p < 0.0001) and LFS (p < 0.0001).
- The continuous and categorical models for the IPSS-Molecular provided a significant improvement in model discrimination across the three endpoints (LFS, OS, and AML-t) compared with IPSS-R.
Patient restratification
Mapping of the IPSS-R to the IPSS-Molecular using 5-5 category mapping (by combining the moderate–high and moderate–low categories in IPSS-Molecular) showed that 46% of patients were restratified.
- 34% of patients were upstaged, while 12% were downstaged.
- 7% of patients were shifted by ≥2 risk strata.
- >50% of those in the IPSS-R intermediate risk group were restratified, with 11% upstaged to very high by IPSS-Molecular.
- 54% of patients remained in their original risk strata.
Validation cohort
Despite enrichment in the higher-risk patients with MDS, there was an equally good separation across the risk strata in terms of LFS (p < 0.0001) and OS (p < 0.0001), and a significant increase in C-index for model discrimination (LFS, OS, and leukemic transformation; compared to IPSS-R). The proportion of patients that were restratified from the IPPS-R strata were also validated.
Generalizability of IPSS-Molecular
Historically, male patients with MDS have been shown to have worse clinical outcomes compared with female patients as they have an enrichment for genes that are associated with higher risk. However, the IPSS-Molecular was shown to be equally applicable to males and females; therefore, sex is not a cofounder for the IPSS-Molecular stratification.
In general, patients with therapy-related MDS had worse IPSS-Molecular scores, and an enrichment in very high- and high-risk groups compared to those with de novo MDS. However, there was no difference between the observed trajectories in terms of OS and LFS between the two groups.
Conclusion
The IPSS-Molecular clinical decision support tool (calculator) has been developed to enable the incorporation of user-specific variables for each patient, including their unique clinical and molecular features, to generate a patient-specific risk score. The IPSS-Molecular is the first model to deliver a generalizable and reproducible personalized risk score, and is applicable across gender and MDS types.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

